Life Sciences
Turning Scientific Innovation Into Real-World Impact
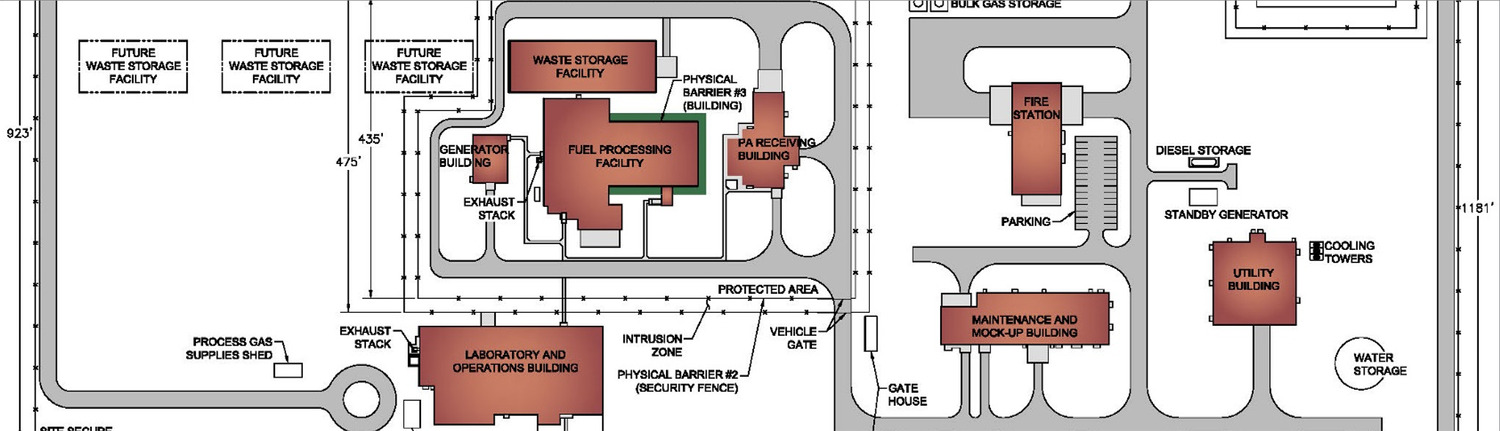
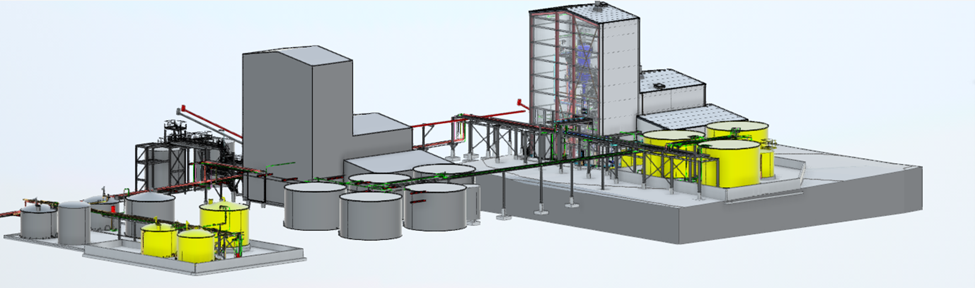
Life sciences organizations operate at the edge of innovation, where science, safety, and operational realities must align. Merrick partners with clients delivering real-world outcomes—including human and veterinary diagnostic laboratories, pharmaceutical and biomanufacturing facilities, and specialized radioisotope and containment environments. We design facilities that support regulated, mission-critical operations where reliability, biosafety, and throughput matter as much as discovery. Our teams translate complex scientific and operational goals into environments that perform as intended from day one, helping clients advance human, animal, and environmental health at scale.
What sets Merrick apart is our fully integrated team of architects, engineers, scientists, commissioning agents, biosafety professionals, and facility operators. Rather than applying isolated disciplines, we solve problems holistically. This allows us to design environments that reflect how science actually happens, not just how it looks on paper. From flexible lab planning to containment strategies and operational workflows, we account for real-world use that traditional firms often overlook.
Regulatory compliance is a baseline expectation in life sciences. Merrick’s value goes further. Our teams help clients optimize space, improve operational efficiency, and reduce lifecycle costs — often fitting more capability into existing footprints. We also help shape the industry itself. Merrick professionals contribute to the development of governing codes and standards in the U.S., Canada, and the UK, giving clients confidence that their facilities meet both current and emerging requirements.
From national laboratories and academic institutions to public health and healthcare interfaces, Merrick supports life sciences missions that demand rigor, adaptability, and long-term thinking.
Why Merrick
- An integrated team of scientists, engineers, architects, and operators solving problems collaboratively
- Deep experience designing facilities for science that have never been done before
- Life-cycle support from planning and funding through design, licensing, and transition
- Expertise that goes beyond compliance to improve operational and cost efficiency
- Active contributors to life sciences codes and standards in multiple countries
- Trusted partner on complex, long-term programs with lasting public impact
- Deep expertise navigating regulatory approvals, supporting successful licensure with agencies such as PHAC, CFIA, CCAC, CDC, NIH, and AAALAC