Medical Isotopes
Engineering The Facilities & Systems Producing Life-Saving Radiopharmaceuticals
Medical isotope production sits at the intersection of nuclear science and pharmaceutical manufacturing, where reliability, speed, and regulatory compliance directly affect patient access and commercial success. Merrick partners with commercial isotope producers to design GMP‑compliant facilities and production environments that safely and consistently deliver radioactive drug products for clinical and commercial use. By combining deep nuclear engineering expertise with life sciences and pharmaceutical facility design, we help clients bring products to market and scale with confidence.
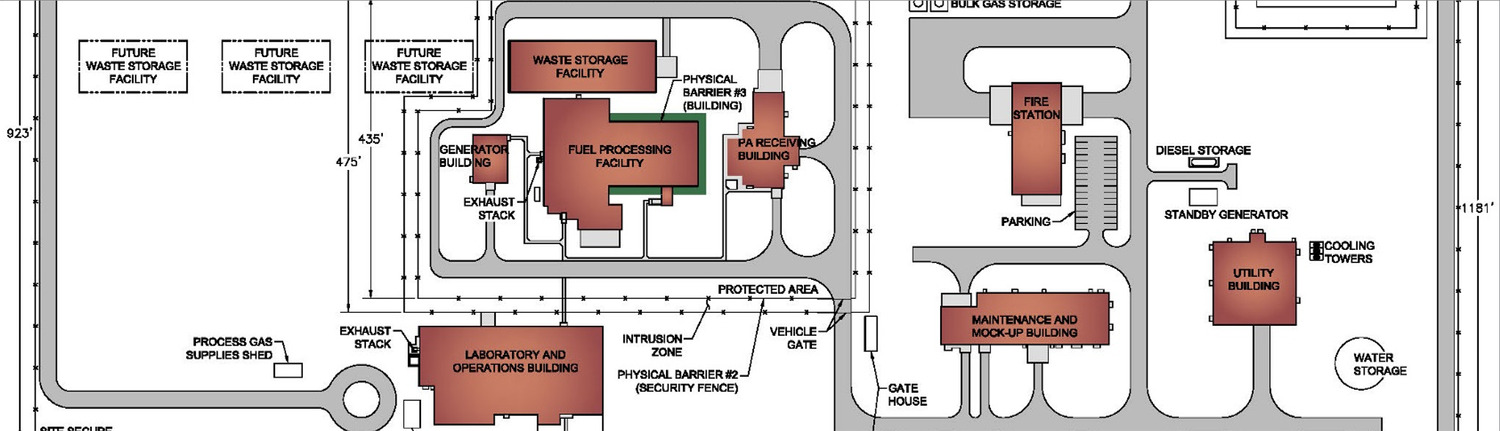
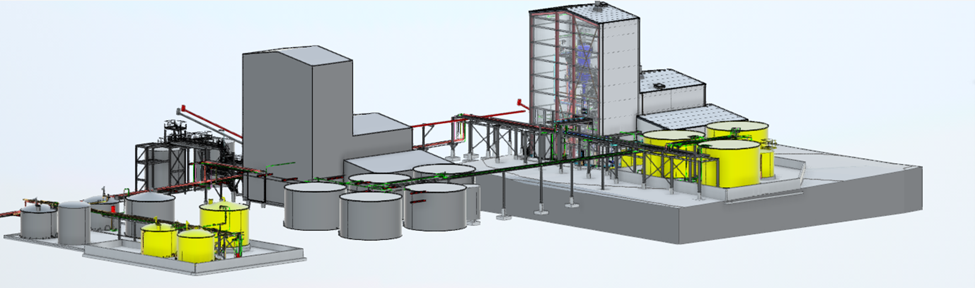
Unlike traditional pharmaceutical manufacturing, medical isotope production must accommodate radioactive materials with extremely short half‑lives—often measured in hours or days. These constraints demand highly reliable operations, efficient material flow, and precise process timing. Merrick designs aseptic, GMP‑aligned production environments—including hot cells, shielded enclosures, and supporting utilities—that enable predictable throughput while protecting workers, the public, and product quality from production through distribution.
Commercial isotope facilities operate within a uniquely complex regulatory environment. In addition to FDA and current Good Manufacturing Practice (cGMP) requirements, producers must comply with Nuclear Regulatory Commission (NRC) and Department of Transportation (DOT) regulations governing radioactive materials. Merrick’s experience delivering medical isotope facilities designed to FDA, EU, and GMP standards supports smoother inspections, reduces regulatory risk, and helps avoid costly redesigns that can delay commercialization.
Merrick’s integrated facility and equipment design approach is a critical differentiator. By engineering building systems, nuclear confinement, and GMP production environments together, we reduce interface risk, shorten delivery schedules, and support long‑term operational reliability. This unified approach enables isotope producers to ramp up production efficiently, expand capacity over time, and maintain consistent supply as demand grows—ensuring facilities are built not just to start up, but to perform throughout the full lifecycle of commercial operations.
Why Merrick
- Integrated facility, equipment, and process engineering accelerates time‑to‑market while reducing cost, schedule, and operational risk for commercial isotope production
- Proven expertise delivering GMP‑compliant, aseptic isotope manufacturing environments, including radioactive APIs and finished drug products, designed for inspection readiness and consistent product quality
- Production‑focused designs optimized for short half‑life isotopes, enabling high throughput, reliable scheduling, and efficient distribution to downstream customers
- Regulatory fluency across FDA/cGMP and nuclear licensing requirements, supporting predictable approvals and smoother transitions from clinical to commercial operations
- Scalable facility solutions that support phased expansion, capacity growth, and evolving isotope portfolios without disrupting ongoing operations
- Experience modernizing existing facilities and delivering purpose‑built commercial production environments, helping operators maximize asset utilization and long‑term return on investment