Biopharmaceuticals & Biotechnology
Facilities That Help Novel Therapies Move From Concept to Clinic
Biopharmaceutical development requires facilities that can adapt to rapidly evolving science while meeting strict regulatory and biosafety requirements. Merrick partners with clients to plan and design biopharmaceutical and biotechnology facilities that support the full development lifecycle—from research and pilot-scale manufacturing to clinical and commercial production. Our multidisciplinary teams translate complex bioprocess needs into environments that support cell, gene, viral vector, and recombinant products.
Many organizations struggle to scale from laboratory discovery to clinical or full-scale manufacturing. Merrick helps bridge that transition by designing flexible facilities that support early-stage production, pilot operations, and full-scale manufacturing within the same strategic framework. These facilities enable clients to maintain control over quality, compliance, and operations as therapies advance, without overbuilding or limiting future expansion.
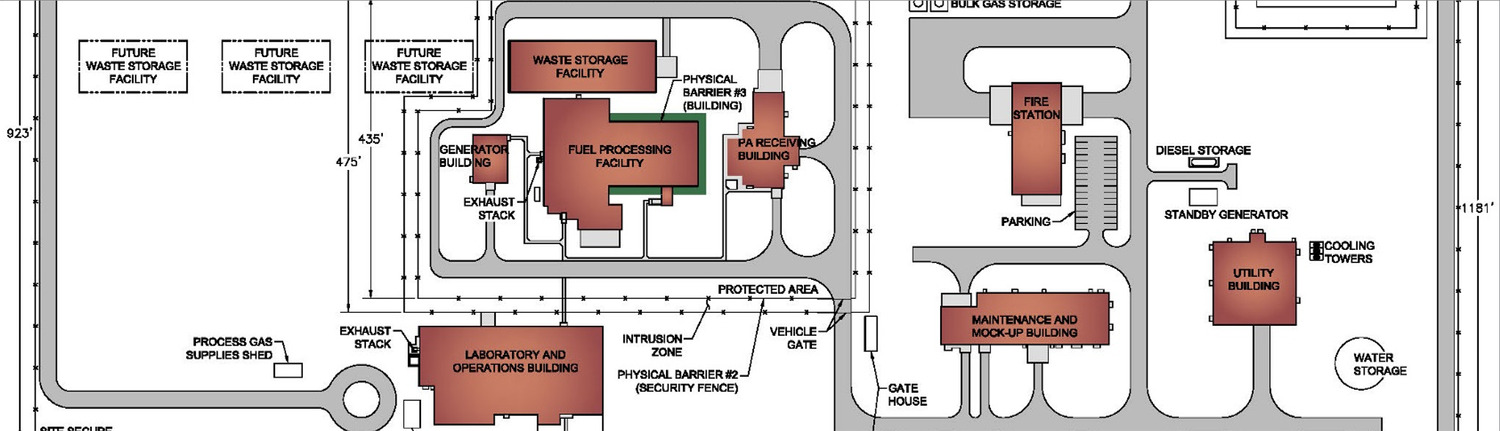
Our role extends beyond design alone. Merrick supports biopharmaceutical facilities through integrated planning, architecture, MEP, fire protection, automation, and controlled environments, with a focus on GMP compliance, biosafety, and operational performance. We design facilities for high-containment production, including work with RG 3 organisms, and support risk analysis, commissioning, and long-term adaptability. Our teams include contributors to governing biocontainment and GMP-adjacent standards in the US, Canada, and the UK.
Why Merrick
- Facility planning and design for pilot, clinical, and full-scale biopharmaceutical manufacturing
- Specialized experience designing high-containment and RG‑3 production environments
- Integrated architectural, MEP, fire protection, automation, and controlled environment expertise
- Support for GMP compliance, biosafety, biosecurity, commissioning, and risk-based decision making
- Scientific insight into advanced bioprocessing and evolving biologics technologies
- Team members who contribute to industry codes and regulatory standards worldwide